10 Best Strategies for IPSC Differentiation in Modern Research?
Induced pluripotent stem cells (iPSCs) have revolutionized regenerative medicine. Their ability to differentiate into various cell types is crucial for advancements in personalized therapy. Leading expert Dr. Karen L. Bullock once said, "The future of iPSC differentiation lies in our understanding of cell signaling pathways." This illustrates the significance of targeted differentiation strategies.
Understanding the intricacies of iPSC differentiation is a complex challenge. Researchers often face hurdles in achieving stable and efficient cell types. Current strategies include optimizing culture conditions and utilizing growth factors. Yet, the outcomes can be inconsistent. Despite the promise of iPSCs, perfection remains elusive in this field.
Innovative thinking is necessary for overcoming these limitations. Each new attempt at iPSC differentiation offers insights, but surprises can arise. The journey is filled with both successes and setbacks, emphasizing the need for continuous learning and adaptation. As science progresses, the quest for reliable methods in iPSC differentiation continues. The road ahead holds potential for significant breakthroughs.

Innovative Approaches for IPSC Differentiation in Modern Research
In modern research, innovative approaches for induced pluripotent stem cell (iPSC) differentiation are shaping new paradigms. One striking technique involves using biomaterials that mimic the stem cell niche. This creates a more natural environment for differentiation.
These materials can provide biochemical cues, altering cell behavior significantly.
Another promising strategy is the integration of small molecules. These compounds can selectively modulate signaling pathways. They influence the fate of iPSCs, pushing them toward specific cell types. Researchers have noted that the timing and dosage of these molecules are crucial.
Mistakes in this phase can lead to inefficient differentiation or unwanted cell types.
The role of 3D culture systems cannot be overlooked. They promote cellular interactions that are vital for appropriate differentiation. However, these systems present challenges in complexity and scalability.
Researchers must refine their methods to ensure reproducibility.
The need for better control over the culture environment remains a hurdle. While progress is evident, the path to optimization requires careful reflection and adjustment.
Key Factors Influencing IPSC Differentiation Strategies
Induced pluripotent stem cells (iPSCs) have revolutionized research, but differentiation remains complex. Various factors play a crucial role in the success of iPSC differentiation. Genetic background is one key aspect. Different cell lines may respond uniquely to differentiation signals, leading to variability in results. Researchers must carefully select their starting materials to ensure consistency.
Another important factor is the culture environment. The composition of the culture medium can significantly influence differentiation outcomes. Specific growth factors and signaling molecules can guide cells toward desired lineages. However, balancing these variables is not always straightforward. Often, optimization requires trial and error. Researchers might face unexpected challenges that lead to suboptimal differentiation.
Moreover, understanding the timing of signal application can impact results. Too early or too late exposure to certain factors can disrupt the process. This adds another layer of complexity. High-throughput screening methods may help identify effective combinations. Yet, not every approach is guaranteed to yield expected outcomes. Continuous reflection on strategies is necessary to refine and enhance iPSC differentiation techniques.
Comparison of Techniques for IPSC Differentiation Effectiveness
Induced pluripotent stem cells (iPSCs) have revolutionized regenerative medicine. Researchers are keen on finding effective differentiation strategies. Each technique has its advantages and drawbacks. Understanding these can lead to better outcomes in research.
When comparing techniques for iPSC differentiation, consider the role of growth factors. Different growth factors promote various cell types. For example, factors like BMPs are crucial for mesenchymal differentiation. However, the timing and concentration of these factors can affect results. It's not always straightforward, and researchers need patience.
Tips: Always test multiple growth factor combinations. It can lead to unexpected results. Don't hesitate to revisit some basic methods. Sometimes, the simplest approach yields the best insight into differentiation.
Another aspect to explore is genetic manipulation. Employing CRISPR or other methods can enhance differentiation efficiency. Yet, introducing genetic changes can present risks. Evaluating these risks is necessary, but it may slow down the research pace.
Tips: Map out your genetic strategy carefully. Collate data on successful protocols and analyze them. Keep track of what works and what doesn’t, as this will guide future experiments. Understanding the fragile balance in these methods will be key to effective differentiation.
Comparison of Techniques for iPSC Differentiation Effectiveness
Challenges and Solutions in IPSC Differentiation Processes
The differentiation of induced pluripotent stem cells (iPSCs) presents significant challenges in modern research. One major issue is achieving reliable and consistent differentiation outcomes. Various protocols exist, but results can vary between experiments. This inconsistency can lead to difficulties when attempting to reproduce findings. Researchers must refine methodologies to enhance reproducibility across different labs.
Another challenge is the time-consuming nature of iPSC differentiation. Traditional methods often require prolonged culture periods. This not only delays research progress, but also increases the risk of cell line variability. Implementing automation and monitoring systems could help streamline processes. Yet, the cost of such technologies poses a barrier for many research facilities.
Moreover, understanding the underlying mechanisms of differentiation remains complex. The signaling pathways involved are not fully understood. Addressing this knowledge gap is crucial. Simplifying the differentiation environment may foster better outcomes. However, it also raises questions about the authenticity of the derived cells. Continuous reflection and adaptation of strategies are essential for overcoming these hurdles in iPSC research.
10 Best Strategies for IPSC Differentiation in Modern Research
| Strategy | Description | Challenges | Solutions |
|---|---|---|---|
| Cell Type Specification | Defining specific cell types to direct differentiation. | Difficulty in maintaining pluripotency during specification. | Use of small molecules to enhance signaling pathways. |
| Growth Factor Optimization | Optimizing growth factors to promote cell differentiation. | Suboptimal growth factor ratios can hinder differentiation. | Conduct extensive tests to find optimal concentrations. |
| Biomaterial Scaffolds | Using scaffolds to support cell growth and differentiation. | Selecting the right materials can be challenging. | Evaluate different biomaterial properties for compatibility. |
| 3D Culture Systems | Implementing 3D culture for more realistic environments. | Complexity in setting up and maintaining 3D cultures. | Utilize commercially available 3D culture kits. |
| Genetic Engineering | Using genetic tools to enhance differentiation. | Potential for off-target effects in gene editing. | Use advanced techniques like CRISPR for precision. |
| Metabolic Reprogramming | Altering metabolic pathways to facilitate differentiation. | Understanding metabolic shifts can be complex. | Utilize metabolomics for comprehensive analysis. |
| Immune Modulation | Adjusting immune factors to favor differentiation. | Could lead to unintended immune responses. | Conduct thorough assays to monitor immune status. |
| Single-cell Analysis | Employing single-cell technologies to track differentiation. | Data complexity and analysis can overwhelm researchers. | Use specialized software for data integration. |
| Customizable Differentiation Protocols | Developing bespoke protocols tailored to specific needs. | Time-consuming and resource-intensive process. | Collaborate with other labs to share insights and protocols. |
Future Directions in IPSC Differentiation Research and Applications
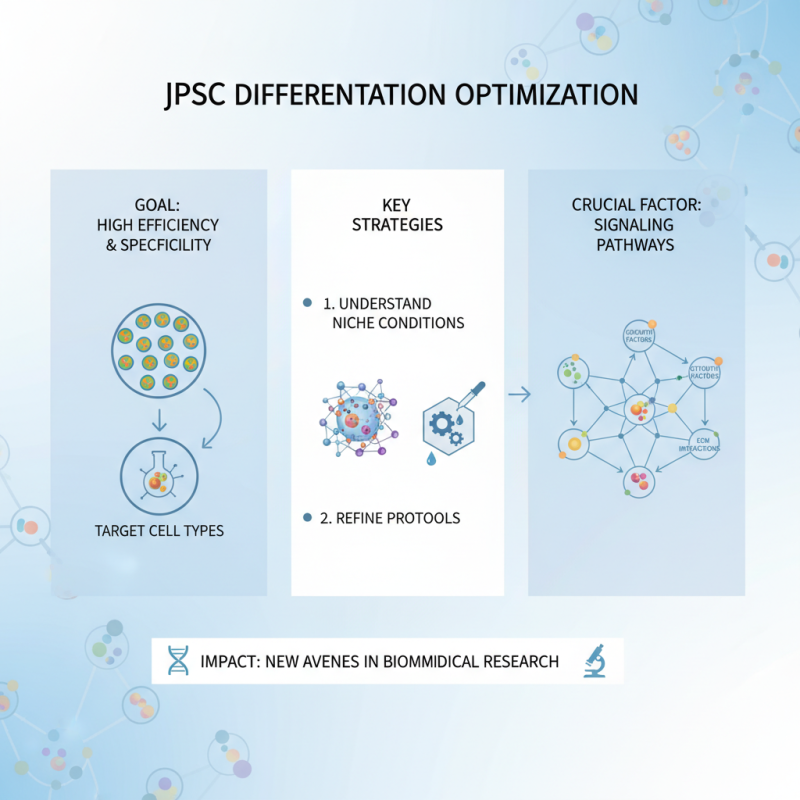
Recent advancements in induced pluripotent stem cell (iPSC) differentiation have opened up new avenues in biomedical research. Researchers are now focusing on optimizing differentiation protocols. They aim for high efficiency and specificity in generating target cell types. Understanding the niche conditions that influence fate decisions is crucial. For instance, examining signaling pathways can refine these protocols significantly.
Emerging technologies play a vital role in this area. 3D culture systems are gaining attention. They better mimic the in vivo environment. Such systems can enhance cell maturation and function. Moreover, single-cell sequencing provides insights into cell heterogeneity. This information is essential to understand and improve differentiation outcomes. However, research is not without challenges. The variability in response among different cell lines can complicate results. Ongoing studies must address these inconsistencies for more reliable applications.
The future of iPSC differentiation lies in personalized medicine. Tailoring approaches to individual patient needs could transform treatment strategies. Continued investigation into the ethical implications is necessary as well. Balancing innovation with responsible usage is key. In this dynamic field, critical reflection will drive meaningful progress.
Related Posts
-
10 Essential Tips for Working with IPSC Cells for Research Success
-

2026 How to Understand Cell and Gene Therapy Advances?
-
How to Understand and Utilize Embryonic Stem Cells in Research?
-

Top 10 Facts You Should Know About Staminal Cells?
-

Why Are Cell Lines Essential for Biomedical Research?
-

10 Best Clinical Research Practices for Successful Trials?
