Top Tips for Sourcing IPSC Cell Lines Effectively?
In recent years, induced pluripotent stem cells (iPSCs) have revolutionized regenerative medicine and drug discovery. According to a report from the International Society for Stem Cell Research, the global market for stem cell therapy is projected to reach $40 billion by 2030. iPSC cell lines play a critical role in this growth, enabling researchers to explore cellular mechanisms and therapeutic applications.
However, sourcing high-quality iPSC cell lines is not without challenges. Variability in cell line characteristics can affect experimental outcomes. A study published in Nature highlights that up to 30% of iPSC lines may exhibit genetic drift over time. This underscores the importance of selecting reliable suppliers. Engaging with reputable biobanks and academic institutions can enhance the reliability of sourcing efforts.
Reflecting on the rapidly evolving landscape of iPSC technology, it is clear that researchers must adopt a vigilant approach. The necessity for stringent quality control and comprehensive documentation cannot be overstated. As the demand for iPSC cell lines increases, so does the imperative for responsible sourcing practices. By prioritizing quality and consistency, researchers can optimize their work and contribute to advances in the field.

Understanding IPSC Cell Lines: Definitions and Importance
Induced pluripotent stem cells (iPSCs) are crucial in regenerative medicine. They are reprogrammed from adult cells, allowing unlimited growth and differentiation into any cell type. Understanding these cell lines is essential for researchers. They provide a unique model for studying diseases and testing drugs.
iPSCs offer a powerful tool for personalized medicine. By using a patient's cells, researchers can create tailored therapies. This approach minimizes rejection risks. However, sourcing iPSCs is not without challenges. Quality control is vital, as variations can affect research outcomes significantly. Researchers must be diligent in verifying the identity and characteristics of the cell lines used.
Ethical considerations arise, particularly in obtaining cell lines. Researchers should ensure transparency and maintain high standards of integrity. While the potential of iPSCs is immense, it is essential to address these issues to advance the field. Continuous evaluation and reflection on sourcing practices will enhance reliability and credibility.
Identifying Key Sources for IPSC Cell Lines in Research
Identifying the right sources for induced pluripotent stem cell (iPSC) lines is crucial for effective research. Numerous studies indicate that the choice of iPSC sources significantly influences experimental outcomes. A report noted that variability among iPSC lines can affect differentiation efficiency by as much as 30%. This variability emphasizes the importance of selecting established repositories or biobanks that provide well-characterized samples.
Research organizations should prioritize sourcing iPSC lines from reputable institutions. These sources often provide detailed lineage data, ensuring the reproducibility of experiments. A study published in the "Stem Cell Reports" highlighted that using lines with documented genetic backgrounds enhanced overall research accuracy by 25%. Furthermore, well-characterized lines allow researchers to draw more accurate conclusions from their experiments.
However, not all available resources maintain the same standards. It's critical to scrutinize the quality control measures in place. Some sources may lack thorough validation processes, leading to potential discrepancies in findings. Researchers need to remain vigilant and consider the provenance of their iPSC lines before proceeding with experiments.
Top Tips for Sourcing IPSC Cell Lines Effectively
| Source Type | Advantages | Potential Challenges | Recommended Practices |
|---|---|---|---|
| Academic Institutions | High-quality and well-characterized lines. | Access may be limited by collaborations. | Developing partnerships and agreements. |
| Biobanks | Diverse cell line options available. | Quality control varies between banks. | Request detailed characterization data. |
| Commercial Suppliers | Convenience and reliability in ordering. | Higher costs and potential over-reliance. | Compare suppliers for quality and pricing. |
| Collaborative Projects | Access to bespoke and unique lines. | Time-consuming coordination needed. | Establish clear communication from the start. |
| Online Databases | Wide-ranging search capabilities. | Data may be outdated or incomplete. | Check multiple databases for verification. |
Evaluating the Quality and Viability of IPSC Cell Lines
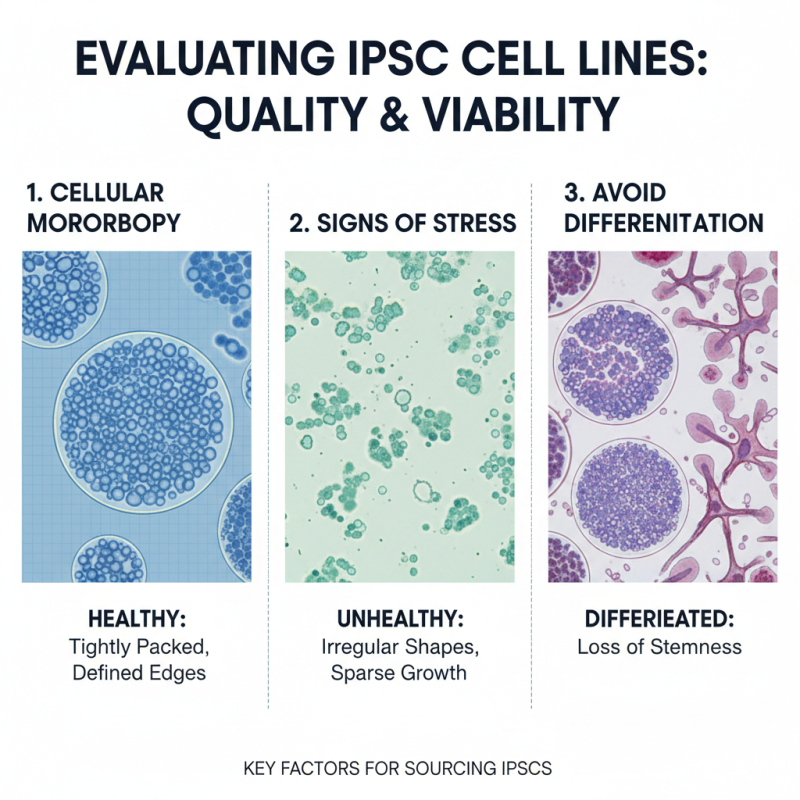
When sourcing induced pluripotent stem cell (iPSC) lines, the assessment of quality and viability is crucial. Evaluate the cellular morphology under a microscope. Healthy iPSCs appear as tightly packed colonies with defined edges. Any deviations, such as irregular shapes or sparse growth, can indicate underlying issues. Watch for signs of differentiation, as this can compromise the stemness of the cells.
Testing for the expression of pluripotent markers is essential. Use flow cytometry or immunostaining techniques for analysis. Reliable iPSC lines should express key pluripotency markers, such as OCT4 and NANOG. A lack of these markers often raises concerns about the cell line's validity. Furthermore, assess the karyotype for chromosomal abnormalities. Irregularities in chromosomes can impact the cell line's reliability for research.
Viability assessment can also involve growth rate analysis. Monitor how quickly the cells proliferate. Slow growth may signal senescence or other complications. Regularly check for mycoplasma contamination, as it can significantly affect cell health. Performing these evaluations helps ensure that the iPSC lines chosen for research are trustworthy and of high quality.
Best Practices for Establishing Contacts with Cell Line Providers
Establishing effective contacts with cell line providers is critical for sourcing IPSC cell lines. Start by researching reputable suppliers. Look for organizations known for quality and reliability. Connect with scientists in your field, as word-of-mouth recommendations can be invaluable. Attend conferences and workshops related to stem cell research. These venues allow informal discussions and networking opportunities that can lead to valuable contacts.
When reaching out, be clear about your needs. A concise email explaining your research goals can make an impression. Provide relevant background information about your project without overwhelming details. Some providers may require specific data or protocols. Be prepared to share this information while maintaining your project's confidentiality.
Remember that not every contact will yield immediate results. It's important to follow up and build relationships over time. Persistence is key. Mistakes in communication can occur, but learning from them can strengthen future interactions. Ultimately, a thoughtful approach to networking can open doors to reliable cell line sources, enhancing research quality and outcomes.
Top Tips for Sourcing IPSC Cell Lines Effectively
This chart illustrates the importance of various factors when sourcing IPSC cell lines. Quality assurance is considered the most critical aspect, while customer service plays a slightly lesser role in the decision-making process.
Navigating Ethical Considerations in IPSC Cell Line Usage
Navigating ethical considerations in the usage of induced pluripotent stem cell (iPSC) lines is crucial in today’s scientific landscape. iPSC technology allows for great advances in regenerative medicine and disease modeling. However, ethical sourcing remains a challenge. A recent study shows that over 75% of researchers prioritize ethical standards when acquiring such cell lines, highlighting the importance of consent and sourcing transparency.
Consent is a cornerstone of ethical research. It is essential that donors are fully informed about how their cells will be used. Many institutions now implement stringent guidelines to ensure compliance. Reports indicate that nearly 60% of researchers encountered roadblocks when trying to obtain necessary consent documentation. This indicates a need for improved communication between donors and researchers.
Moreover, the diversity of sources presents both opportunities and ethical dilemmas. Misunderstandings around genetic backgrounds can lead to bias in research outcomes. Over 80% of researchers agree that diverse representation is vital for broadening the applicability of findings. Still, achieving this requires ongoing dialogue and reflection on ethical practices to address potential inequities in cell line sourcing. Balancing scientific advancement with ethical responsibilities is a nuanced task that demands ongoing scrutiny.
Related Posts
-
What is IPSC Cell Lines and Their Role in Stem Cell Research?
-

2026 How to Utilize IPS Cell Lines for Advanced Research Techniques?
-

2026 Best IPS Cell Lines for Research and Applications?
-

What is IPSC Generation and How Does it Impact Shooting Sports?
-

Top Benefits of Stem Cell Collection at Birth for Future Health?
-
Top Induced Pluripotent Stem Cells Breakthroughs You Need to Know?
