Top Insights on IPS Cell Lines You Need to Know?
Induced pluripotent stem (iPS) cell lines have revolutionized regenerative medicine and research. These versatile cells can differentiate into various cell types. Their potential applications are vast, from disease modeling to drug discovery. However, the journey of iPS cell lines is not without challenges. Ensuring their safety and stability is paramount.
Researchers have made strides in improving the efficiency of iPS cell generation. Yet, the long-term effects of these cells remain under investigation. This uncertainty highlights the need for ongoing studies. They can provide critical insights into genetic stability and potential risks. Understanding these factors is essential for their translation into clinical applications.
In this landscape, reliable iPS cell lines must be prioritized. Available cell lines may vary in quality and performance. Researchers must consider these factors when selecting a line for their studies. Collaborative efforts in the scientific community can enhance the development and accessibility of high-quality iPS cell lines. Overall, awareness and understanding of these complexities are necessary for advancing the field.

What Are Induced Pluripotent Stem (IPS) Cells?
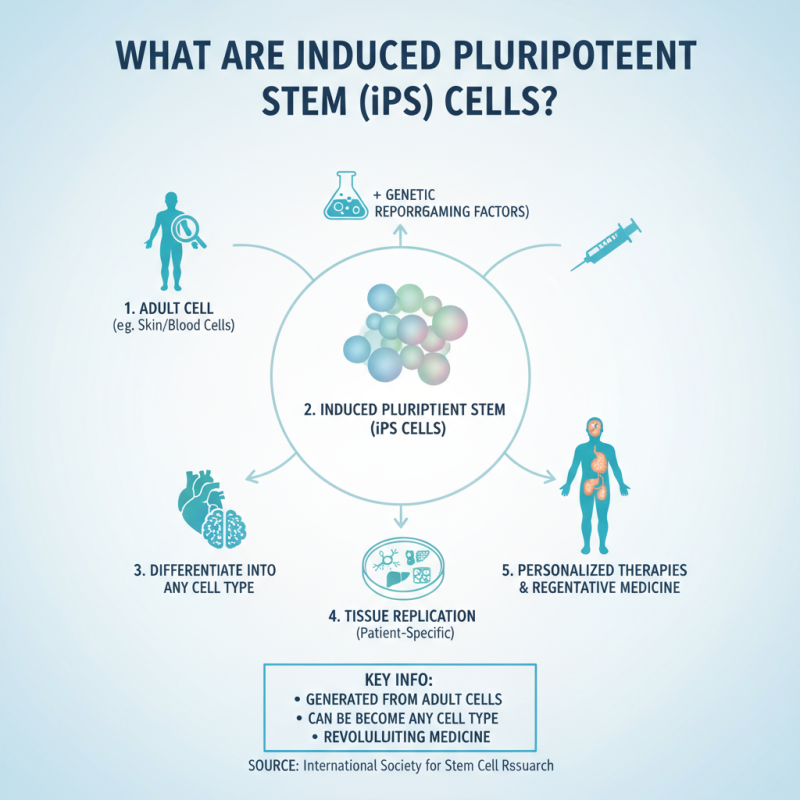
Induced pluripotent stem (iPS) cells are a type of stem cell that can be generated directly from adult cells. These cells have the ability to differentiate into any cell type, making them a valuable asset in regenerative medicine. According to the International Society for Stem Cell Research, iPS cells can replicate various patient-specific tissues, which opens up new avenues for personalized therapy.
Research indicates that iPS cells are not without challenges. They can carry genetic abnormalities, affecting their reliability. A recent study revealed that approximately 20-40% of iPS cell lines show mutations that could influence cell behavior. This raises concerns about their use in clinical settings. Furthermore, the differentiation process must be carefully controlled to ensure the development of functional cells, as improper conditions can lead to incomplete or dysfunctional tissues.
Despite potential drawbacks, the impact of iPS cells is profound. They have already contributed to significant breakthroughs in drug testing and modeling diseases. Reports suggest that over 50 clinical trials involving iPS cells are currently underway globally. The need for rigorous quality control and standardized protocols remains crucial in harnessing their full potential for therapeutic applications.
Historical Development and Breakthroughs in IPS Cell Research
Induced pluripotent stem (iPS) cells have revolutionized the field of regenerative medicine. The historical development of iPS cell research dates back to 2006 when Shinya Yamanaka introduced the groundbreaking technique. This discovery allowed somatic cells to be reprogrammed into pluripotent stem cells, capable of differentiating into any cell type. Yamanaka's work earned him the Nobel Prize in Physiology or Medicine in 2012, highlighting the potential of iPS cells for both clinical and research applications.
Since then, iPS technology has made significant strides. For instance, a 2021 report indicated that over 600 clinical trials involving iPS cells were in progress worldwide. These trials span a range of diseases, including Parkinson's disease and diabetes, showcasing the versatility of iPS cells in addressing various health challenges. Researchers now grapple with issues related to cell stability and long-term efficacy. While progress is notable, challenges remain in ensuring reliable and consistent cell behavior after differentiation.
Moreover, the ethical considerations surrounding iPS cell research emphasize the importance of informed consent and patient privacy. Although iPS cells avoid some ethical dilemmas linked to embryonic stem cells, the potential for misuse in genetic modification persists. As the field evolves, rigorous standards must be maintained to ensure the responsible application of these powerful tools in medicine. The ongoing quest for safety and effectiveness remains a critical aspect of iPS cell research.
Key Characteristics and Advantages of IPS Cell Lines
Induced pluripotent stem (iPS) cell lines have revolutionized regenerative medicine and drug discovery. These cells are reprogrammed from somatic cells, which allows them to differentiate into any cell type. This property makes them invaluable for disease modeling and therapeutic applications. Research indicates that the global iPS cell market is projected to grow significantly, reaching approximately $2.3 billion by 2025, reflecting a strong demand for these innovative tools in biomedical research.
One key characteristic of iPS cells is their pluripotency. This enables researchers to generate any cell type, such as neurons or cardiomyocytes, from a single source. A study published in Nature Biotechnology noted that iPS cell lines provide a renewable source of cells, which is essential for long-term studies. Furthermore, they minimize ethical concerns compared to embryonic stem cells. However, challenges remain, including issues like genetic stability and potential tumorigenicity. Researchers must continually assess these risks, ensuring that iPS cells can be safely used in clinical settings.
The adaptability of iPS cells is another major advantage. They can be derived from various tissues, customizing cell lines to specific patient needs. For example, they can be created using skin or blood samples. This personalization drives progress in personalized medicine, allowing for targeted therapies tailored to individual genetic profiles. Despite these benefits, scientists must focus on standardizing protocols to enhance reproducibility. Ongoing investigations aim to refine these methods, addressing inconsistencies in cell quality and functionality.
Applications of IPS Cells in Medicine and Regenerative Therapy
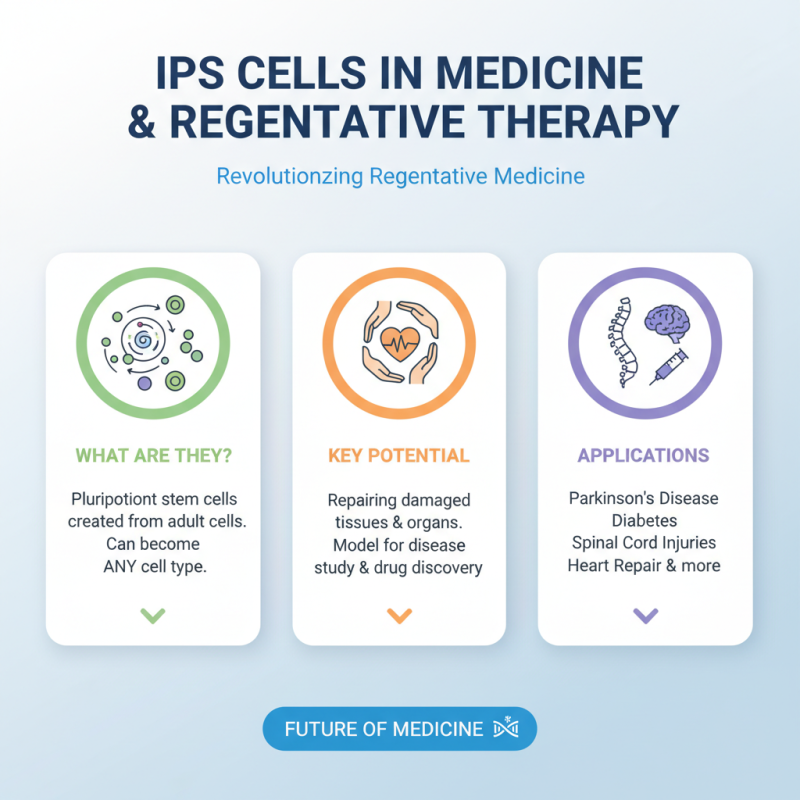
Induced pluripotent stem (iPS) cells are revolutionizing regenerative medicine. These cells offer a unique ability to differentiate into any cell type, providing strong potential for therapies. Recent studies indicate that iPS cells could play a pivotal role in treating degenerative diseases and injuries. For instance, they are being investigated for applications in Parkinson's disease, diabetes, and spinal cord injuries.
The deployment of iPS cells in regenerative therapy has sparked significant interest. A report from the International Society for Stem Cell Research (ISSCR) highlights that over 75% of clinical trials involving stem cells utilize iPS technology. This illustrates their growing prominence in the medical field. However, challenges remain, particularly regarding safety and long-term effects. There are instances of tumorigenesis associated with iPS cell therapies, raising important questions about their use.
Tip: When considering iPS cells for research or therapy, consult peer-reviewed studies and engage with trusted experts in the field. Always prioritize safety and ethical standards. Additionally, staying informed about the latest advancements is crucial as the landscape of regenerative medicine evolves rapidly. Maintaining a critical eye on emerging data and experimental outcomes can enhance understanding and foster responsible innovation.
Challenges and Future Directions in IPS Cell Line Development
Induced pluripotent stem (iPS) cells hold immense promise for regenerative medicine. However, developing stable iPS cell lines poses significant challenges. One major issue is the variability in differentiation potential among different iPS lines. These differences can lead to unpredictable outcomes in research and therapy. Inconsistent cell growth rates also complicate the establishment of standardized protocols. This inconsistency can result in difficulties when comparing results across studies.
Moreover, the risk of genetic mutations during reprogramming adds another layer of complexity. These mutations can influence the behavior of the cells, impacting both their utility and safety in clinical applications. Researchers must also navigate ethical considerations while striving for reproducibility in iPS development. Despite widespread enthusiasm, there is a pressing need for clarity in regulatory guidelines related to the use of these cells.
Future directions involve improving the reprogramming techniques to enhance efficiency and consistency. Employing sophisticated genome editing methods may help remove undesired mutations. There is hope that advanced bioinformatics can aid in monitoring cellular changes over time. Embracing a more standardized approach to iPS cell line development may ultimately bolster the field’s credibility and reliability.
Related Posts
-

Tips for Choosing the Best Stem Cell Banks for Your Needs
-
10 Essential Tips for Working with IPSC Cells for Research Success
-
10 Essential Tips for Working with Pluripotent Stem Cell Lines
-

10 Best Cell Stem Cell Innovations Transforming Medicine Today?
-

Top 10 Facts You Should Know About Staminal Cells?
-

Why is IPSC Generation Important for Competitive Shooting?
