10 Essential Tips for Working with Endothelial Cell Lines
Endothelial cell lines play a crucial role in vascular research and disease modeling. These cells line the blood vessels and are essential for understanding cardiovascular health. Working with endothelial cell lines requires precision and knowledge. Researchers face unique challenges in cell growth and maintenance.
Optimal conditions for culturing these cells vary. Each endothelial cell line has specific requirements. Factors such as temperature, medium composition, and substrate influence cell behavior. It is vital to tailor protocols to individual cell lines. This approach maximizes their potential for experimental applications.
Collaboration with experts can enhance research quality. Engaging with experienced peers helps overcome common pitfalls. Researchers should remain adaptable and open to feedback. Each experiment offers a chance to learn and improve techniques. Embracing a continual improvement mindset can lead to significant advancements in using endothelial cell lines.

Understanding Endothelial Cell Lines: Importance and Applications
Endothelial cell lines play a crucial role in biomedical research. They help scientists understand vascular biology. These cells line blood vessels, acting as a barrier between circulating blood and tissues. Their study can illuminate how blood flow influences inflammation and tissue health. Researchers frequently use these cell lines to uncover disease mechanisms, particularly in cardiovascular and cancer research.
While working with these cells, researchers must ensure that they maintain the right culture conditions. Endothelial cells require specific growth factors and a controlled environment. Variability in these factors can lead to inconsistent results, which can be frustrating. It is essential to monitor cell morphology and growth patterns regularly. Observing changes can indicate potential issues with the culture environment.
Moreover, researchers should explore the genetic differences between various endothelial cell lines. Not all endothelial cells behave the same way. Some may be more suitable for specific experimental conditions. Understanding these nuances can improve experimental design and outcomes. Embracing these complexities in endothelial cell research can lead to deeper insights into vascular health and disease.
Choosing the Right Endothelial Cell Line for Your Research Needs
Choosing the right endothelial cell line is crucial for your research. Different endothelial cell lines have unique characteristics that impact their behavior. Some lines mimic human vasculature closely, while others may not. Understanding these differences allows researchers to select the best fit for their experiments.
Consider the source of the endothelial cells. Lines derived from specific tissues may exhibit distinct properties. Human umbilical vein endothelial cells (HUVEC) are a common choice. However, they might not represent all types of endothelial cells. Alternatively, brain-derived lines can be more relevant for neurovascular research. It's essential to weigh the advantages of each line against your specific research goals.
Pay attention to the growth conditions as well. Some cell lines require unique media formulations or specific growth factors. Failure to provide the right environment can lead to poor results. Experimentation and optimization may be necessary to achieve the desired cell function. Be prepared to troubleshoot and adapt. This adaptability will enhance your research outcomes significantly. Choosing wisely is key, but so is learning from the process.
10 Essential Tips for Working with Endothelial Cell Lines
Best Practices for Culturing Endothelial Cell Lines Effectively

Culturing endothelial cell lines can be a rewarding yet challenging task. To succeed, one must consider various factors such as cell line selection and environmental conditions. Maintain a sterile environment to prevent contamination. This practice significantly reduces the risk of unwanted microbial infestation, which can compromise your results.
Temperature and media composition play crucial roles. Endothelial cells thrive at specific temperatures, usually around 37°C. Adjust the media to suit the specific needs of the cell line. Regularly check the pH and osmolarity. Many researchers overlook these details, leading to suboptimal cell growth.
Another important aspect is passaging techniques. Over-trypsinization can damage the cells, affecting their viability. Aim to use gentle methods for subculturing. Keeping track of the cell's passage number helps monitor their growth characteristics. Tracking these metrics is vital for replicating results reliably. Various factors can influence outcomes, and reflecting on past experiences improves future experiments.
Optimizing Experimental Conditions for Endothelial Cell Line Studies
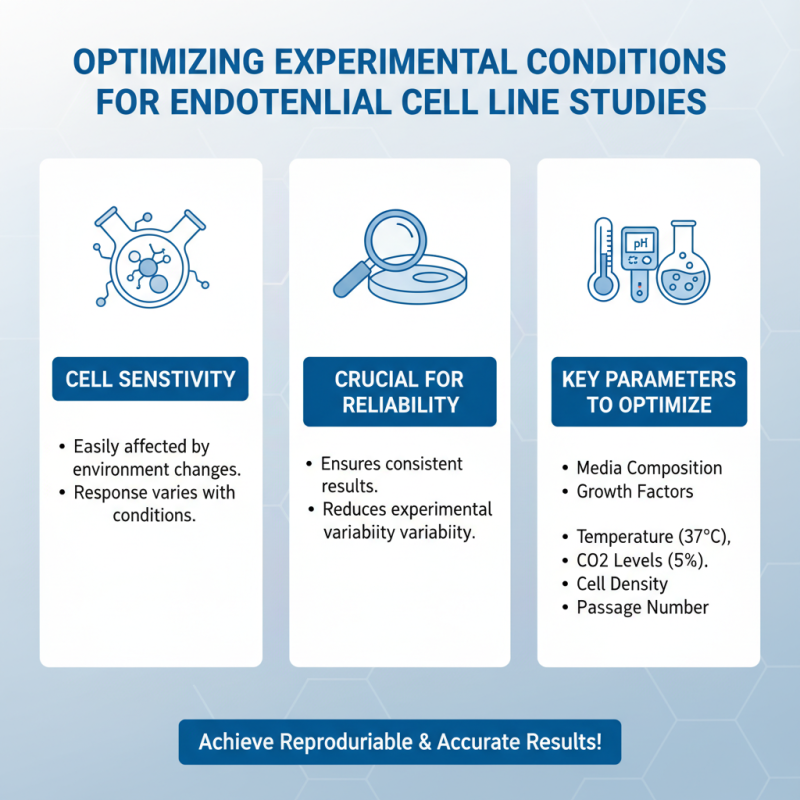
Working with endothelial cell lines requires careful attention to experimental conditions. These cells are sensitive and can behave differently under varying environments. Thus, optimizing these conditions is crucial for reliable results.
One tip is to maintain consistent temperature and CO2 levels. Endothelial cells thrive in a stable environment. Fluctuations can lead to variability in cell behavior. Another important aspect is the choice of culture medium. Ensure it meets the specific nutritional needs of the cells. The medium should support growth and function, promoting optimal results in experiments.
Consider the substrate on which the cells are cultured. Some materials can influence cell adhesion and morphology. Using coated plates may improve cell performance. Finally, regular monitoring of cell health is essential. Cells can deteriorate without visible signs, impacting your findings. Addressing these factors can enhance the reliability of your endothelial cell line studies.
Common Challenges and Troubleshooting Tips in Endothelial Cell Research
Working with endothelial cell lines can present various challenges. One common issue is maintaining cell viability during prolonged culture. Ensure that you monitor the pH levels and media composition regularly. This can prevent cellular stress and enhance growth rates.
Another challenge is the differentiation of endothelial cells into various phenotypes. Different conditions can yield unexpected results. Adjusting growth factors can help in achieving desired outcomes. Remember to document any changes in conditions and their effects on cell behavior. This can provide insights for future experiments.
Furthermore, contamination is a persistent threat in any cell culture. Strict aseptic techniques are vital. Regularly inspect your cultures for signs of contamination. If contamination occurs, assess the source carefully. It may reveal issues in your laboratory environment or handling practices. Engaging in open discussions with colleagues can provide valuable perspectives and troubleshooting tips. Sharing experiences helps with understanding common pitfalls better.
10 Essential Tips for Working with Endothelial Cell Lines - Common Challenges and Troubleshooting Tips in Endothelial Cell Research
| Tip | Description | Common Challenges | Troubleshooting Steps |
|---|---|---|---|
| Maintain Sterility | Always work in a sterile environment to avoid contamination. | Contamination from bacteria or fungi. | Use disinfectants and work in a laminar flow hood. |
| Use Appropriate Media | Select media that is optimized for endothelial cells. | Poor growth or health of cell lines. | Check for media validity and expiration. |
| Control Temperature | Maintain cell cultures at 37°C. | Cells may not grow properly. | Use calibrated incubators with alarms. |
| Monitor pH Levels | Ensure the pH of media is suitable (around 7.2-7.4). | Media becoming acidic can harm cells. | Regularly measure and adjust pH as necessary. |
| Subculture Frequently | Passage cells before they reach confluence. | Overcrowding can lead to senescence. | Set a schedule for subculturing based on cell growth rate. |
| Optimize Cell Density | Inoculate with the correct number of cells for optimal growth. | Too few or too many cells may affect experiments. | Test various densities to find the optimal range. |
| Avoid Genetic Drift | Regularly check phenotypes to ensure stability. | Alterations can lead to unreliable data. | Store and use frozen vials to maintain consistency. |
| Use Proper Cryopreservation | Freeze cells with optimal cryoprotectants. | Cell loss during freezing and thawing. | Follow standard protocols for cryopreservation. |
| Regularly Assess Cell Health | Confirm viability and morphology regularly. | Poor health can compromise experiments. | Use assays to measure cell viability and morphology. |
Related Posts
-
Top Endothelial Cell Lines for Research and Applications?
-

2026 Top IPSC Cell Lines for Advanced Research and Applications?
-

10 Best Strategies for IPSC Differentiation in Modern Research?
-
Top Induced Pluripotent Stem Cells Breakthroughs You Need to Know?
-
2026 How to Use Stem Therapy for Enhanced Healing and Wellness?
-

2026 Best IPS Cell Lines for Research and Applications?
