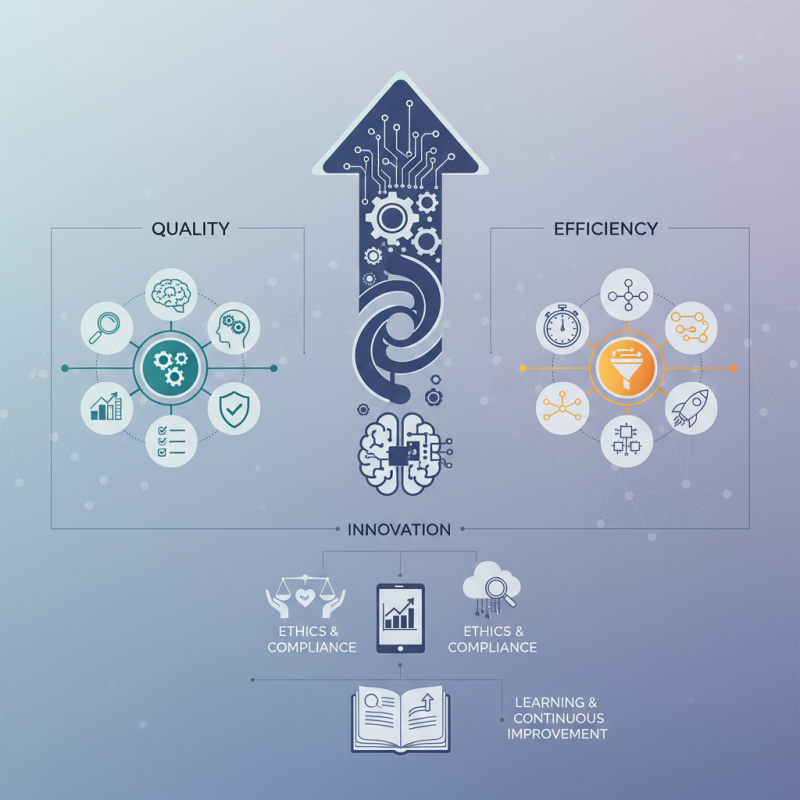
How to Enhance Clinical Research Quality and Efficiency?
Clinical research plays a vital role in advancing medical science and improving patient outcomes. The quality and efficiency of clinical trials directly affect the credibility of findings and their application in healthcare. Enhancing these aspects is essential for stakeholders, including researchers, healthcare providers, and patients.
A focus on quality control can lead to more accurate data collection and analysis. However, achieving this requires careful planning and execution. Researchers must rely on robust methodologies and thorough training for all team members. Efficient processes streamline workflows, reducing delays and costs. Yet, the challenge remains to balance speed and rigor in clinical research.
Innovation is key in this evolving landscape. Technologies like artificial intelligence and big data analytics show promise in improving outcomes. Nevertheless, ethical considerations and regulatory compliance must not be overlooked. Reflecting on past trials can provide invaluable lessons, ensuring future studies are more reliable and impactful. In this complex field, continuous improvement is not just beneficial—it's essential for meaningful progress.
Strategies for Improving Data Management in Clinical Research
Efficient data management is crucial in clinical research. Organizations must focus on streamlining processes to ensure quality. This starts with robust data collection techniques. Utilizing electronic data capture systems can enhance accuracy and mitigate errors. However, some researchers may resist new technology, fearing it complicates workflows. Proper training can address these concerns and encourage adoption.
Data monitoring plays a significant role as well. Regular audits help identify discrepancies early. Minor issues can escalate into significant problems if not addressed promptly. Moreover, integrating data from various sources can enrich research outcomes. Yet, this can lead to challenges in data harmonization and standardization. Researchers need to continually refine their approaches to tackle these complexities.
Collaboration among teams fosters a culture of shared responsibility. Utilizing cloud-based platforms allows real-time data sharing, enhancing communication. However, this raises concerns about data security and access control. Striking a balance is essential. Continuous evaluation of the strategies in place can lead to improved efficiency and reliability in data management. Addressing flaws and learning from them is key to progress.
Enhancing Patient Recruitment and Retention for Clinical Trials

Enhancing patient recruitment and retention is vital for successful clinical trials. According to clinical research reports, nearly 80% of trials fail to meet their recruitment targets on time. This inefficiency delays important discoveries and increases costs. Effective strategies, such as targeted outreach and community engagement, must be implemented to improve these metrics.
Utilizing digital tools can significantly aid in patient recruitment. A study from the Society for Clinical Research Sites revealed that online platforms can double participant enrollment rates. Additionally, providing clear information about trial benefits and processes can reduce patient hesitance. Many potential participants feel overwhelmed or confused about the involvement, which leads to dropouts.
Retention during trials is equally crucial. Research indicates that 30% of patients discontinue participation due to poor communication. Regular updates and transparent feedback loops can address this issue. Furthermore, personalized support for patients can foster loyalty and commitment. Addressing patient concerns effectively contributes to overall study integrity and data reliability, highlighting the need for continuous improvement in recruitment and retention initiatives.
Implementing Innovative Technologies for Research Efficiency
Innovative technologies play a critical role in enhancing the quality and efficiency of clinical research. According to a 2023 report published by the Clinical Trials Transformation Initiative, implementation of digital tools can reduce trial timelines by up to 30%. Electronic data capture and remote monitoring are becoming standard practices. These technologies streamline data management and improve patient engagement.
Mobile health applications enable real-time data collection. They empower patients to report their symptoms and side effects promptly. This leads to more accurate data and better patient compliance. Yet, not all researchers are adopting these tools. A survey conducted by the Bioethics Research Institute found that only 40% of clinical trials are utilizing mobile technology. The hesitation often stems from concerns about data privacy and regulatory uncertainties.
Furthermore, artificial intelligence is transforming data analysis. Machine learning algorithms can identify trends that human researchers might miss. A study by the Journal of Biomedical Informatics revealed that AI could potentially reduce data analysis time by 50%. However, there's still a learning curve for researchers to adapt to these technologies. Continuous training is essential for maximizing benefits and ensuring reliable outcomes. The potential for increased efficiency is clear, but challenges persist in fully realizing these innovations.
How to Enhance Clinical Research Quality and Efficiency? - Implementing Innovative Technologies for Research Efficiency
| Technology | Application | Benefits | Challenges |
|---|---|---|---|
| Electronic Data Capture (EDC) | Streamlining data collection | Increased accuracy and reduced data entry time | Implementation costs and training requirements |
| Telemedicine | Remote patient consultations | Improved patient access and convenience | Technical issues and data privacy concerns |
| AI and Machine Learning | Data analysis and predictive modeling | More accurate and faster results | Data quality and algorithm bias |
| Patient Portals | Patient access to personal health information | Increased patient engagement and satisfaction | User adoption and technology literacy |
| Blockchain | Secure data sharing for clinical trials | Enhanced data security and integrity | Regulatory acceptance and scalability |
Establishing Rigorous Quality Control Practices in Research Processes
Establishing rigorous quality control practices in clinical research is essential for improving outcomes. A recent report from the Food and Drug Administration (FDA) highlighted that nearly 30% of clinical trials face major protocol deviations. Such deviations can lead to skewed data and compromised results. Implementing strict quality control checkpoints can mitigate this risk. This includes auditing processes and routine assessments that ensure adherence to protocols.
Moreover, a study from the Journal of Clinical Research indicated that research sites with robust quality control measures experienced a 40% reduction in data discrepancies. This reflects the strong link between effective oversight and enhanced data integrity. It is vital to have trained personnel conducting regular reviews. Training programs that focus on quality standards can significantly decrease human error, which accounts for a significant percentage of issues in research.
However, despite these efforts, challenges persist. Many researchers overlook minor discrepancies during data collection. These small errors can accumulate, impacting the overall findings. Continuous education about quality practices should not be neglected. Addressing these gaps is crucial for maintaining high standards in clinical research. Effective measures can be integrated, but constant vigilance is necessary.
Enhancing Clinical Research Quality and Efficiency
This bar chart illustrates the efficiency ratings across various stages of clinical research, highlighting the critical areas for enhancing quality control practices.
Fostering Collaboration Among Stakeholders in Clinical Research
Collaboration among stakeholders is critical in enhancing the quality of clinical research. Recent data from the Clinical Trials Transformation Initiative indicates that multidisciplinary collaboration can improve trial outcomes by up to 30%. This collaboration often involves sponsors, investigators, patients, and regulatory bodies. Each group plays a unique role in driving research forward.
Engaging patients in the research design process is particularly impactful. The Patient-Centered Outcomes Research Institute emphasizes that studies with patient involvement are 40% more likely to meet recruitment goals. This highlights the importance of addressing patient needs and preferences from the start. Yet, many trials still lack adequate patient representation, which can lead to skewed results.
Sharing data across stakeholders is also essential. A report from the Tufts Center for the Study of Drug Development found that open data sharing could lead to savings of billions in research costs. However, trust issues often hinder this collaboration. Stakeholders need to foster open communication to build that trust. Integration of diverse perspectives can result in more robust findings, yet many researchers are still hesitant to engage fully with all stakeholders.
Related Posts
-

10 Best Clinical Research Practices for Successful Trials?
-
Why are IPSC Cell Lines Important for Stem Cell Research?
-

What is Stem Cell Collection at Birth and Why is it Important?
-

Why is IPSC Generation Important for Competitive Shooting?
-
Top Stem Cell Types and Their Applications in Medicine?
-

Tips for Choosing the Best Stem Cell Banks for Your Needs
